India is preparing for a nationwide human papillomavirus (HPV) vaccination campaign aimed at reducing cervical cancer, one of the leading causes of cancer deaths among women in the country. As the rollout approaches, public discussion has again surfaced around vaccine safety, past controversies, and the role of global health actors including concerns linked to Bill Gates–funded programmes.
A review of available scientific literature, regulatory findings, and fact-checks shows broad global confidence in HPV vaccine safety, while also highlighting why some public scepticism continues to persist.
Why the Vaccine Is Being Prioritised
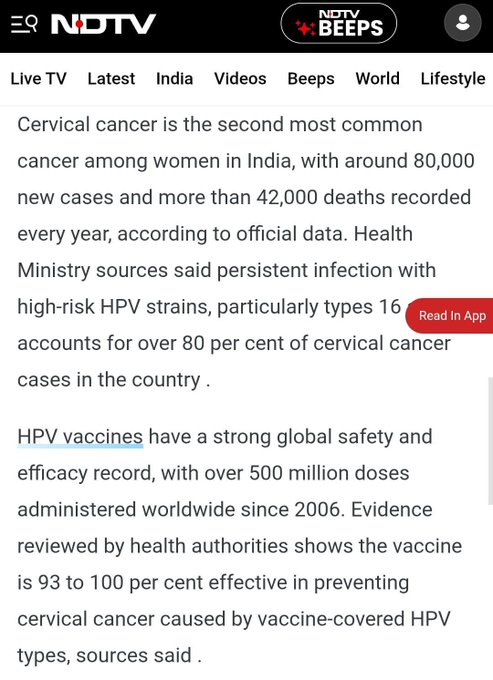
Cervical cancer remains a major public health burden in India. According to health data cited in multiple reports, tens of thousands of new cases and over 40,000 deaths are recorded annually.

Persistent infection with high-risk HPV strains, particularly types 16 and 18, accounts for the majority of cervical cancer cases.
Public health experts say vaccination before exposure to the virus significantly reduces future cancer risk, which is why most national programmes target adolescent girls.
What Long-Term Safety Studies Say
Large international studies over more than a decade have generally found HPV vaccines to have a strong safety profile.
A registry-based cohort study in Sweden and Denmark involving nearly one million girls did not find evidence linking the quadrivalent HPV vaccine to autoimmune, neurological, or venous thromboembolism conditions.

Reviews of long-term safety data have reported that serious vaccine-related adverse events are rare and broadly comparable to other widely used vaccines.
The most commonly reported side effects remain mild injection-site reactions such as pain, swelling, and redness.

The World Health Organization’s Global Advisory Committee on Vaccine Safety has repeatedly reviewed global data and has stated that HPV vaccines continue to be considered extremely safe, noting that anaphylaxis occurs at roughly 1.7 cases per million doses – a very low rate.

Global usage has been extensive, with hundreds of millions of doses administered worldwide since the mid-2000s.
The 2009 India Controversy Explained
Much of the continuing public concern in India traces back to a 2009 HPV demonstration project supported by PATH, a public health nonprofit partly funded by the Gates Foundation.
Seven deaths among vaccinated girls were reported during that period. However, subsequent reviews found:
Five deaths were due to clearly unrelated causes such as drowning, snake bite, malaria, and pesticide exposure.
The remaining two deaths were considered unlikely to be linked to the vaccine.
The Indian Council of Medical Research suspended the project at the time, and a parliamentary committee later raised concerns about procedural and ethical lapses in how the demonstration programme was conducted. Importantly, those findings focused largely on consent and trial processes rather than establishing a causal safety signal for the vaccine itself.

Health authorities note that the proposed national rollout is structurally different from that earlier demonstration exercise.
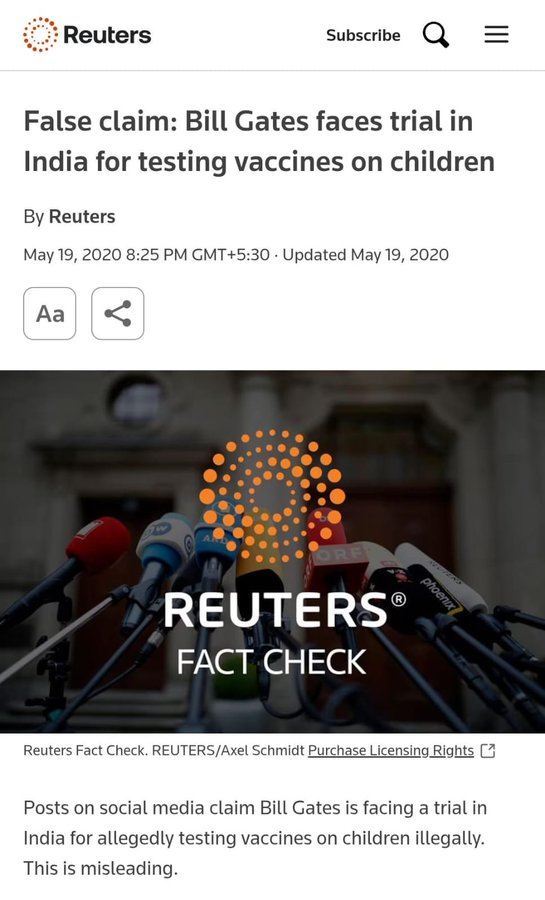
Claims About Bill Gates and Legal Cases
Social media claims have periodically alleged that Bill Gates or his foundation face legal action in India over HPV vaccination. Independent fact-checks, including by international news agencies, have found no evidence of any ongoing court case in India on this issue.

The Gates Foundation has historically funded global immunisation initiatives, including support to organisations involved in HPV programmes, which partly explains why the topic continues to attract public scrutiny.
Effectiveness and Regulatory Status
HPV vaccines have been licensed in India since 2008. International evidence suggests high effectiveness, often cited in the 90–100% range, in preventing cervical cancers caused by vaccine-covered HPV strains when administered before exposure.
India’s planned expansion is expected to follow existing regulatory approvals, WHO recommendations, and structured public-health protocols, including training of healthcare workers and informed consent procedures.
Why Some Scepticism Persists
Public health specialists say vaccine hesitancy around HPV in India stems from multiple factors:
- lingering memory of the 2009 programme controversy
- broader distrust of pharmaceutical companies
- concerns about adolescent vaccination
- misinformation amplified on social media

Experts note that addressing these concerns transparently, rather than dismissing them, will be critical for public confidence.
The Bottom Line
Available global and Indian evidence to date indicates that HPV vaccines have a strong safety and effectiveness record, and major health agencies continue to recommend their use to prevent cervical cancer.
At the same time, past procedural controversies and the involvement of international philanthropic funding have contributed to ongoing public questions in India.
As the nationwide rollout approaches, health authorities face a dual challenge: expanding protection against a preventable cancer while also maintaining public trust through clear communication, monitoring, and transparency.
(This article is based on an X Thread By PoliticalKida)
Subscribe to our channels on WhatsApp, Telegram, Instagram and YouTube to get the best stories of the day delivered to you personally.




